Building Narrative, Credibility and Presence in AI Drug Discovery
Context
Scientists are trained to be skeptical.
When an AI company walks into a room full of researchers, regulatory professionals, and drug developers promising transformation, the response can sometimes be suspicion instead of excitement.
The life sciences audience has heard the disruption pitch before, so leading with it risks making storytelling and positioning harder, not easier.
Having spun out Form Bio from Colossal Biosciences (the de-extinction company bringing back the wooly mammoth, dire wolf and dodo), I knew we needed to earn credibility with people who don't give it easily.
What I Did
I architected an ambitious, multifaceted program assortment to rapidly build the reputation and position of Form Bio within the biopharma industry.
While I ran a complete marketing mix appropriate for our industry and commercial model, there were 5 specific pillars I took on that were particularly consequential:
01 - Turning scientific leaders into visible and credible industry voices
02 - Assembling a PhD-level scientific marketing supergroup
03 - Harnessing scientific advisory and academic relationships that helped AI feel safer for skeptical researchers
04 - Being seen and heard with industry leaders like NVIDIA, Google, PacBio, and Ginkgo Bioworks
05 - Building proximity to the people and conversations shaping the regulatory landscape
The details of these pillars follow:
01 - Turning scientific leaders into visible and credible industry voices
Form Bio's Chief Strategy Officer, Dr. Claire Aldridge, had deep expertise across immunology, genetics, and tech transfer—but no public profile in biopharma technology and AI-driven drug discovery.
To expand and shape her industry profile, we built a systematic placement program to get Claire seen and heard in the media and on the stages that mattered.
Select Placements:
BiotechTV · Form Bio's Chief Strategy Officer on optimizing the payload of cell and gene therapies
PharmaVoice · Woman of the Week
SynBioBeta · Cell & Gene Manufacturing
Molecule to Market · The puzzle-solving, lifelong science geek
Genetic Engineering News · Cell Therapy Manufacturing Grapples with Scale-Up
American Society of Gene & Cell Therapy · Utilizing AI in Gene and Cell Manufacturing
02 - Assembling a PhD-level scientific marketing supergroup
Credibility with scientific audiences requires content, messaging and material developed by people who've actually done the science.
I recruited a team of PhD-level scientific marketers—former bench scientists with research backgrounds across life sciences fields.
Here's our all-star team, who I still maintain relationships with:
Jill R / Content Strategist
PhD in Immunology, Tufts
Bijan Z / Product Marketing
PhD in Biochemistry, Oxford
Elie D / Copywriter
PhD in Biomedical Engineering, UCSB
Tina B / Copywriter
PhD in Biology, University of Konstanz
Alice Y / Social Media
PhD in Biomedical Informatics, Stanford
Patrick W / SEO
Masters in Chemistry, University of Hull
Selected content developed by our team:
Regulatory Thought Leadership
Analysis of emerging FDA guidance and its implications for gene therapy developers.
Examples:
What the FDA’s New Safety Draft Guidance for AAV Gene Therapies Means and How to Be Ready
Design for Manufacturing: A New Paradigm for Computational Life Sciences
Scholarly Publications
Peer-reviewed research co-authored with scientific advisors and partners.
Examples:
Developing Machine Learning Powered Solutions for Cell and Gene Therapy Candidate Validation
CRISPRank: A Tool for Determining the Best CRISPR Genome Editing Method
Mini Case Studies
Quantified proof points showing AI/ML solving specific AAV vector design problems.
Examples:
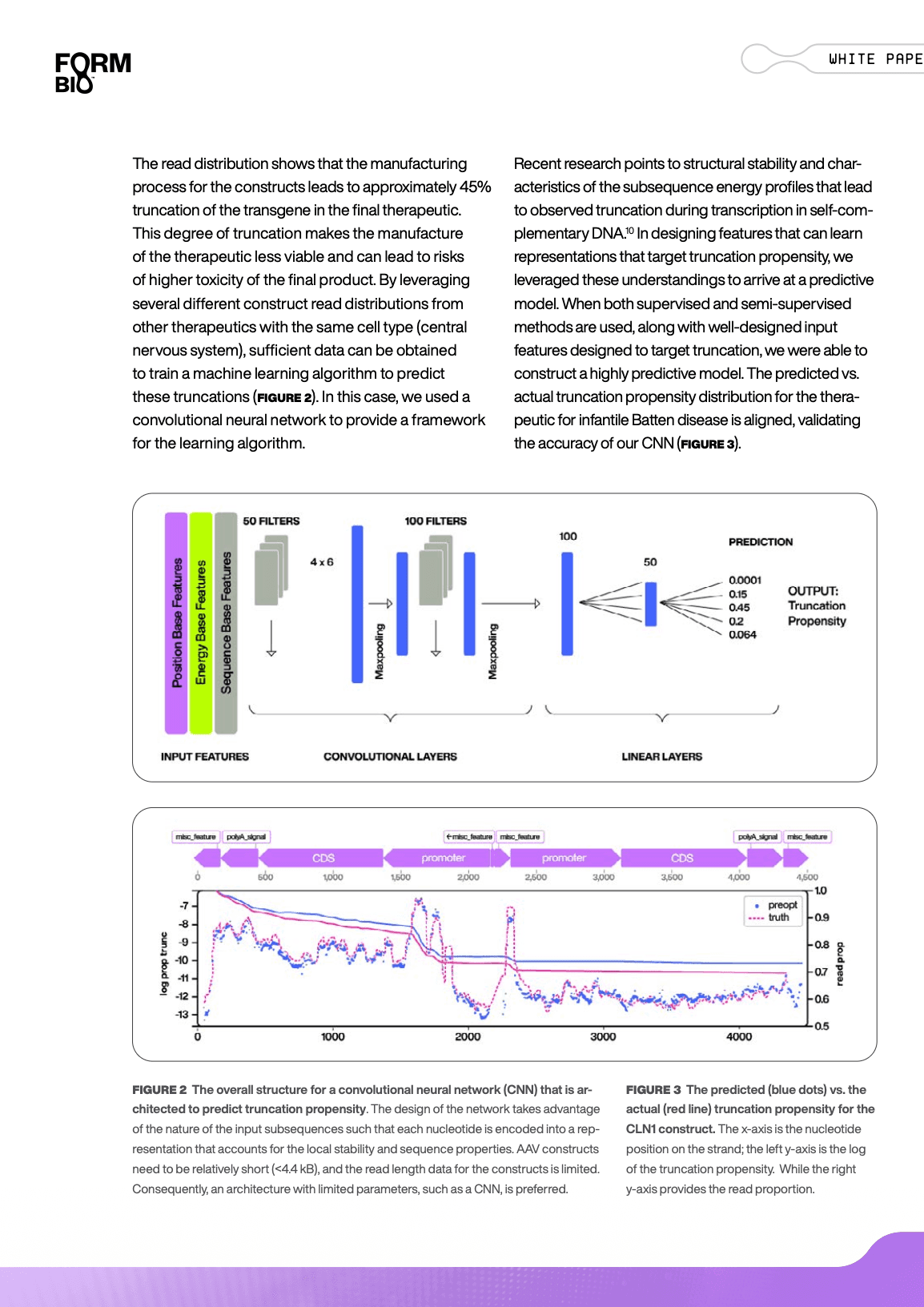
AI/ML Optimization of Coding Sequences Reduces Genome Truncations in AAV Vectors
Reducing the Risk of Immunogenicity Using an AI/ML Model
AI/ML Optimization Reduces AAV Discovery Timeline from Years to Days
Webinars
Live scientific discussions with partner scientists and executive leaders from Google Cloud, NVIDIA and the biopharma R&D community.
Examples:
Data Science Meets Life Science: AI Challenges & Opportunities in Biopharma, Synthetic Bio & Beyond
Explore Google’s Advanced Omics AI Models and Key Discoveries with Form Bio
03 - Harnessing scientific advisory and academic relationships that helped AI feel safer for skeptical researchers
Form Bio's (and Colossal's) scientific advisors included Nobel Laureate Carolyn Bertozzi (2022 Chemistry, Stanford) and George Church (Harvard/MIT, pioneer of the Human Genome Project), alongside leading researchers in cell and gene therapy from prestigious academic and corporate labs.
These relationships represented a variety of research partnerships, several of which we harnessed for marketing publications, community events, and speaking opportunities.
Our academic relationships (and how we presented them) provided an important dimension of credibility and made AI feel safer and more accessible for skeptical researchers in both industry and academia. Our advisory and academic relationships played a critical role in helping us teach the market how AI and established scientific process come together.
04 - Being seen and heard with industry leaders like NVIDIA, Google, PacBio, and Ginkgo Bioworks
NVIDIA
Highlighted on stage at GTC by both Jensen Huang and Kimberly Powell (VP Healthcare). Co-published a joint case study. Selected as a reference platform integration for Parabricks accelerated genome sequencing.
Example:
Mammoth Mission: How Colossal Biosciences Aims to ‘De-Extinct’ the Woolly Mammoth
Google Cloud
Selected as the exclusively recommended multiomics platform for Google's healthcare, life sciences, and academic segments. Featured in Google's own Multiomics Suite launch announcement as a co-launch partner.
Example:
Form Bio, Google Partner on Cloud-Based Multiomic Analysis Software
Ginkgo Bioworks
Named Ginkgo's inaugural Technology Network partner for AAV gene therapy. CEO Jason Kelly personally quoted calling Form Bio "a pioneer" in the joint announcement.
Example:
Form Bio’s AI Ensures Gene Therapies Pass Muster, with Ginkgo’s Help
PacBio
Selected as the exclusively recommended computational solution for PacBio's gene therapy segment.
Example:
PacBio, FormBio Unifying AAV Industry, Insilico Medicine Introduces New Products, More
Each of these partner activations required coordinated PR, joint content, co-marketing execution, and careful brand positioning.
The comms strategy is what turned technical integrations into visible, high-signal endorsements.
05 - Building proximity to the people and conversations shaping the regulatory landscape
JPM Healthcare Conference
At the annual JPM Healthcare Conference, the Super Bowl of biopharma dealmaking and partnering, we leveraged our ARM relationship to align Form Bio's brand with Dr. Peter Marks, then-Director of the FDA's Center for Biologics Evaluation and Research—the most influential single regulator for advanced therapies.
Exclusive Community Events
We created an intimate leadership dinner series across the U.S. and featuring respected industry luminaries including Tim Hunt, CEO of the Alliance for Regenerative Medicine (ARM)—the foremost industry body for cell and gene therapy before regulators and policymakers.
Validated Regulatory Efficiency
By leveraging partner FDA submissions showing minimal questions and pushback on IND filings, we were able to move the industry toward standardization (for filing data packages) for AI-designed drug candidates.
Regulatory Thought Leadership
Through our scientific content publishing efforts like the ones mentioned earlier, we positioned the company as one that understood the regulatory environment and could provide significant advantages in navigating regulatory process.
Outcome
Form Bio rapidly converted spinout attention into scientific credibility.
Our content, our executive visibility, our academic and industry affiliations and our hosted events track collectively positioned Form Bio as a credible player in the space.
This category authority directly supported Form's commercial pipeline, with customers like Bristol Myers Squibb, Solid Biosciences and Cure Rare Disease entering long term, 8-figure partnerships with us.